
Antigen retrieval (HIER or PIER) is mainly required for formalin-fixed paraffin-embedded tissues, since fixation and embedding cause strong protein cross-linking that masks epitopes. In fixed but non-paraffin tissues, such as cryostat or vibratome sections, retrieval is often not necessary because the fixation is milder. However, some epitopes may still be masked, and certain antibodies work better with retrieval. A good approach is to first test staining without retrieval, then apply HIER or PIER if signals are weak.
HIER is the most used antigen retrieval method and involves heating tissue sections in a buffer to reverse formalin-induced cross-linking. Instruments for Heat-Induced Epitope Retrieval (HIER) include various heat sources like microwaves, pressure cookers, food steamers, and water baths, and specialized automated HIER machines. Each instrument requires optimization of time, temperature, and buffer depending on the tissue type and antigen.
The choice of AR buffer significantly impacts antigen recovery. The most frequently used buffers are:
* Addition of Tween-20 is recommended for paraffin-embedded sections. For non-paraffin sections avoid or minimize Tween-20.
** Tris-EDTA and EDTA buffers enhance sensitivity for most antibodies but may increase background staining.
Each buffer has unique properties that affect epitope exposure. A small-scale test run with different buffers can help determine the optimal condition for specific antibodies.
Proper control of temperature and retrieval time is critical as overheating can lead to tissue detachment, antigen degradation, or increased background staining, while underheating may result in incomplete retrieval weak or absent staining.
Most IHC-P protocols from SYSY Antibodies recommend heat-induced epitope retrieval (HIER) at 97–100°C for 30 minutes using a food steamer (Figure 1), which provides consistent and standardized antigen retrieval conditions.
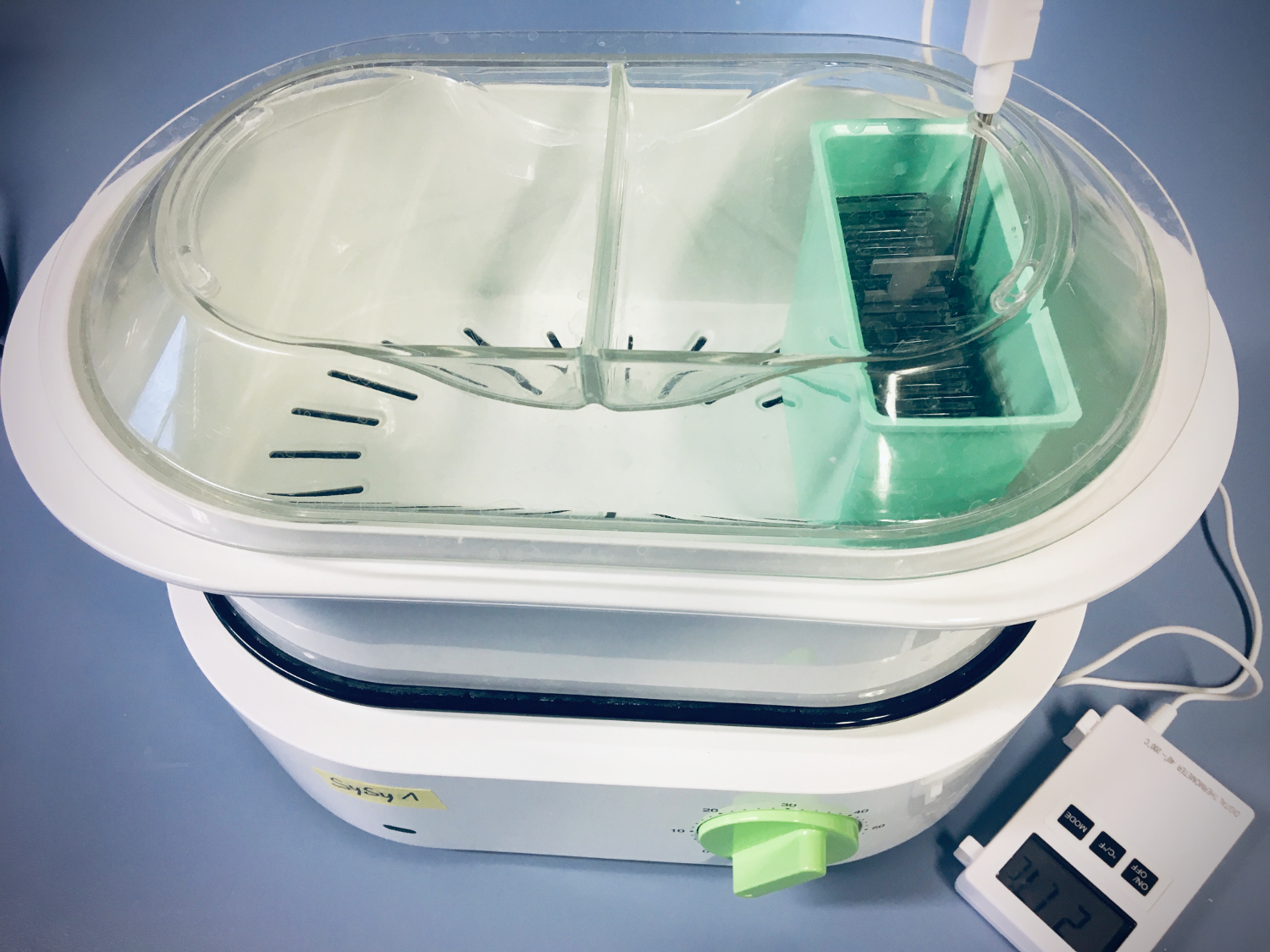
In contrast, fixed tissue sections that are not embedded in paraffin are typically incubated overnight at 60 °C in AR buffer using a water bath. The use of lower temperatures and AR buffer without Tween reduces tissue damage. Conditions should be optimized for each type of tissue and antigen to achieve the best staining results. After HIER, cooling the sections to room temperature helps to stabilize the antigen conformation and prevents abrupt structural changes.
HIER is highly sensitive to pH, time, temperature, and buffer composition (Figure 2).
Since no universal antigen retrieval method exists, we recommend empirical optimization of HIER conditions—including buffer selection, pH, and incubation time—for each antibody and tissue type.
Figure 2: Staining intensity is influenced by antigen retrieval time and temperature. Indirect immunostaining of formalin fixed paraffin embedded (FFPE) mouse spleen sections with upper row: rabbit anti-CD11c (cat. no. HS-375 003, dilution 1:250, DAB, brown) and lower row: rat anti-CD8a (cat. no. HS-361 017, dilution 1:500, DAB, brown). Slides were heated in citrate buffer pH 6.1 (S1699, Agilent) for 20 min or 40 min in a heat steamer or for 5 min in a pressure cooker. Nuclei have been visualized by hematoxylin staining (blue).
PIER involves enzymatic digestion using proteases such as Proteinase K, Trypsin, or Pepsin, typically incubated at 37°C for 10–30 minutes. Enzyme concentration, incubation time, and temperature must be optimized to prevent over-digestion and tissue damage. PIER is preferred for antigens that do not respond well to HIER and can also be combined with HIER for complex antigen retrieval needs (Figure 3). While PIER can enhance antigen exposure, it may compromise tissue morphology and has generally a lower success rate for restoring immunoreactivity.
Figure 3: Influence of PIER and HIER methods on indirect immunostaining of formalin fixed paraffin embedded (FFPE) mouse cerebellum sections with guinea pig anti-Collagen type IV (cat. no. 462 004, dilution 1:500, DAB, brown). The combination of HIER and PIER significantly improves the staining result. A: PIER (20 min 0.2 mg/ml Pepsin (37°C)). B: HIER, pH 9 (30 min 1 mM Tris-EDTA pH 9.0 (97°C)). C: HIER, pH 6 + PIER (30 min 10 mM Citrate pH 6.0 (97°C) + 20 min 0.2 mg/ml Pepsin (37°C)). D: HIER, pH 9 + PIER (30 min 1 mM Tris-EDTA pH 9.0 (97°C) + 20 min 0.2 mg/ml Pepsin (37°C)). Nuclei have been visualized by hematoxylin staining (blue).
Antigen retrieval with formic acid is a special method used in IHC, mainly to improve the staining of amyloid and prion proteins in formalin fixed tissues. Tissue sections are treated with highly concentrated formic acid, which breaks down the amyloid fibrils and improves accessibility for antibodies. In FFPE tissues, formic acid treatment can be combined with HIER to improve staining for amyloid deposits (Figure 4).
Figure 4: Influence of formic acid (FA) treatment on indirect immunostaining for amyloid deposits. Formalin fixed paraffin embedded (FFPE) human brain sections from an Alzheimer's patient were stained with mouse anti-Abeta38/40/42/43 (cat. no. 218 211, dilution 1:1000, DAB, brown) following Antigen Retrieval (AR) in Citrate buffer (Agilent, S1699) alone (A), combination of AR and digestion with proteinase K (B) or combination of AR and FA treatment (C). Nuclei have been visualized by hematoxylin staining (blue).